Our Product
The Cytek Northern Lights™ flow cytometer, with its easy-to-use workflow, is a flexible and affordable full spectrum flow cytometry solution for everyday research applications. The Northern Lights instrument incorporates Cytek’s powerful Full Spectrum Profiling™ (FSP™) technology in a one to three laser system, allowing for a wide array of new fluorochrome combinations without reconfiguring your system for each application. Whether you are doing basic assays such as cell proliferation, apoptosis analysis, or other cell health applications, or multicolor immunophenotyping panels, the Northern Lights system meets all of your flow cytometry needs.
SpectroFlo® software provides a user-friendly guided workflow from quality control to data analysis, incorporating tools to make running applications simpler.

Easy, Flexible, and Intuitive
Low Cost of Ownership
Remarkable Sensitivity
High Value
Testimonials and Publications
Investigating Sample Heterogeneity
Adapting to Multiple Users Needs
Service Plan Comparison
Service Contract Types
Gold
Includes
- Unlimited Coverage Of Parts, Labor & Travel
- 1 Preventative Maintenance Visit/Year
- Priority Response Time
- Unlimited Phone & Email Support
Silver
Includes
- Limited Coverage Of Parts, Labor & Travel
- 1 Preventative Maintenance Visit/Year
- 1 Fully Covered Repair Visit
- Unlimited Phone & Email Support
- 20% Parts Discount
Bronze
Includes
- 1 Preventative Maintenance Visit/Year
- Unlimited Phone & Email Support
- 10% Parts Discount
- All Other Services Billed At Prevailing Rate
Counting and Viability
The Cytek Northern Lights™ full spectrum cell analyzer, along with the Guava® ViaCount™ reagent assay, offers precise and accurate counting and viability determination for many different cell types. The high sensitivity of the Northern Lights instrument offers excellent resolution of populations that may be missed on conventional systems. The workflow is easy and requires minimal effort to acquire counts and viability of cell populations.

Live and dead Jurkat cells were mixed and stained with the Guava ViaCount reagent. Analysis was performed on the Northern Lights system. Percentages shown are the percent of live and dead cells in each sample.

Jurkat cells were induced into undergoing apoptosis with staurosporine treatment. The induced cells, and the live and dead cell controls, were analyzed on the Northern Lights system. The Guava Nexin® assay was used to determine each population. Clear distinction of each population was achieved as shown.
Apoptosis Analysis
Easily detect and measure cell populations progressing through the apoptotic pathway using the Northern Lights full spectrum flow cytometer and the Guava Nexin® assay. You can determine and differentiate between live, dead, and early apoptotic populations in a quick and simple way. SEE DATA SHEET
Cell Cycle Analysis
The Northern Lights instrument is a powerful tool for flow cytometry cell cycle analysis. Using the Northern Lights system, one can easily determine the discrete phases of the cell cycle with percent CVs well under 3%. Not only can you analyze cell cycle progression, but you can multiplex with phenotyping of important cell cycle protein markers. The beauty of Full Spectrum Profiling™ (FSP™) technology allows for deeper insights into cell division and cell cycle analysis.

The cell cycle inhibitor, nocodazole, was used to arrest cells in the G2/M phase of the cell cycle. Samples were treated, prepared with the Guava® Cell Cycle reagent, and data were acquired on the Northern Lights flow cytometer. FCS files were analyzed using ModFit LT™ software (Verity Software House).
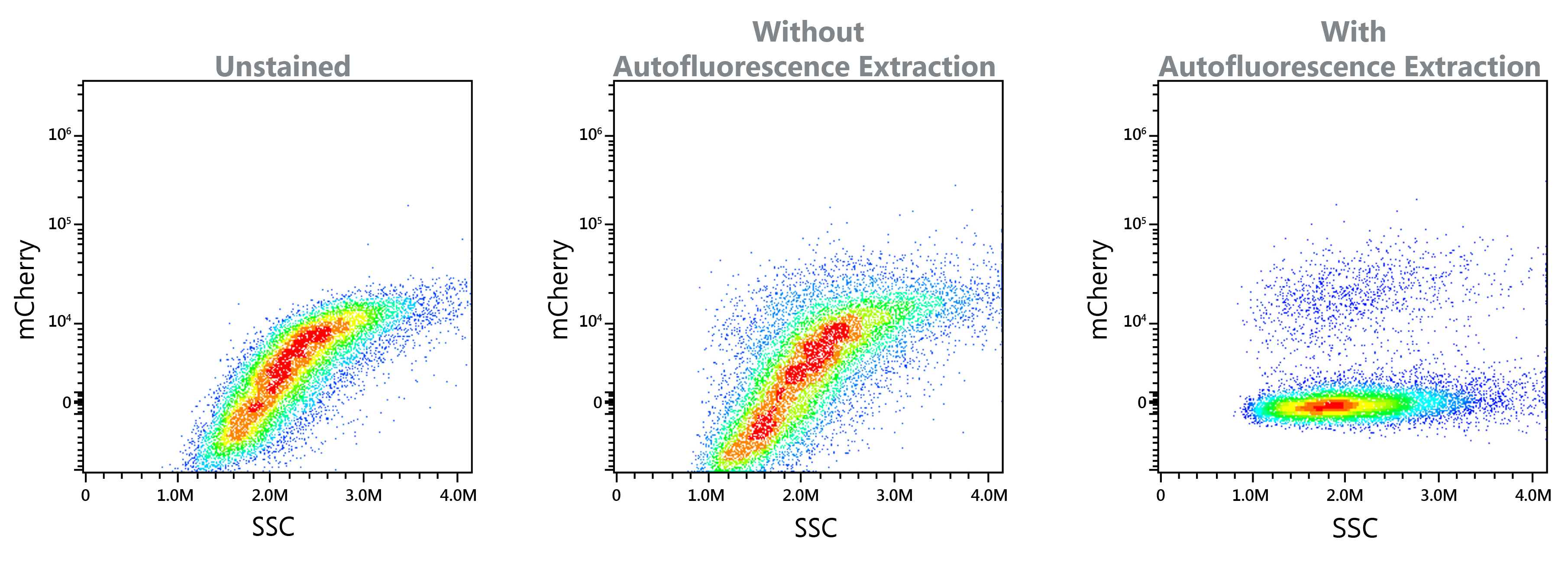
Autofluorescence Extraction
The Northern Light system’s implementation of full spectrum flow cytometry enables the use of autofluorescence extraction to further improve data clarity. Certain sample types, such as yeast and tumor samples, present the challenge of high autofluorescence. For these challenging applications involving highly autofluorescent particles, let the software's autofluorescence extraction tool bring new levels of resolution. SEE POSTER
Everyday Immunoprofiling
9 Colors
The one laser (488 nm) Cytek® Northern Lights system can perform 9-color immunoprofiling assays with no sacrifice to data quality. In this 9-color blue laser excitable dyes panel, monocytes and several CD4 T cell and CD8 T cell subsets were easily identified.

21 Colors
With 30 fluorescence detectors and only 2 lasers (405 and 488 nm), the Cytek® Northern Lights system enables multi-parametric data collection with few lasers to maintain. Cytek has developed a 21-color human immunophenotyping panel acquired from just a single tube, with outstanding resolution.

Class 1 Laser Product.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.





